Tiring struggles three decades soon
2008/12/01 Orive Arroyo, Gorka - Farmazian doktorea. Biofarmazia, Farmakozinetika eta Farmazia-teknologiako irakasle kolaboratzaileaFarmazia Fakultatea UPV-EHU, Vitoria-Gasteiz | Zarate Sesma, Jon - Farmazian doktorea. Biofarmazia, Farmakozinetika eta Farmazia-teknologiako irakasle kolaboratzaileaFarmazia Fakultatea UPV-EHU, Vitoria-Gasteiz Iturria: Elhuyar aldizkaria

Today, 33 million people are affected by AIDS worldwide. However, there are also encouraging data from the 17th International AIDS Congress held in August 2008 in Mexico. Publication of the report prepared by the United Nations Conference, which analyzes data from 147 countries.
For example, compared to previous years, the number of disease-related deaths has decreased. In addition, the percentage of pregnant hiv-positive women receiving antiretroviral treatment to prevent contamination of newborns has increased by 19% between 2005 and 2007. In the same time period, the number of new cases of infection in children has decreased from 410,000 to 370,000. Moreover, more and more countries are taking steps to achieve the universality of antiretroviral therapy. For example, Botswana, Brazil, Chile, Costa Rica, Cuba and Laos are on the verge of achieving therapeutic universality.
In addition, patients' life expectancy has increased considerably since the virus was detected to date. As published in The Lancet last July, the life expectancy of those infected with the virus has elongated 13 years between 1996 and 2005 in Europe and North America.
The research has been led by researchers from the Canadian University Simon Fraser and the Bristol University of the United Kingdom, within the project The Antiretroviral Therapy Cohort Collaboration, and has been based on the follow-up of 43,355 people who at age 20 began receiving antiretroviral therapy. Currently, the life expectancy of patients undergoing treatment is around 70 years of age in the early stages of the disease. The quality of life of people with AIDS has improved greatly and, in general, the society of developed countries is more aware than a few years ago.
Visible drugs
In addition, the rapid evolution of science brings interesting news every day. For example, new strategies for drug development. Antiretroviral therapy has so far been based on preventing the proliferation of the virus. To do this, two enzyme inhibitors, transcriptase and protease inhibitors are used. However, many seropositives develop their resistance to the drugs of these two families, so scientists have searched for new medicines.
One of them is the drug erraltegrabir, which has been on sale in Spain since March 2008. This drug is the first in the family of integrase activity inhibitors, which inhibit the penetration of HIV genetic material into human cells. For the moment, its use is recommended in patients with high virus load and resistant to other drugs.
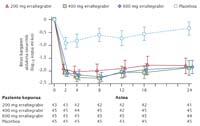
Patients who have received the new drug have shown spectacular results. According to the latest data, after 48 weeks of treatment, erraltegrabir has demonstrated the ability to reduce the virus to undetectable levels. Gradually, the drug is able to increase the number of CD4 cells in the immune system. CD4 cells are T-type lymphocytes that initiate the immune response and destroy HIV. Therefore, the patient is unprotected against other viruses or bacteria.

The next step with the Erraltegrabir is to analyze the result given by the patient as the first treatment option. According to the researchers' hypothesis, raltegrabir can eradicate the virus within six or seven years, which will prevent the patient from undergoing lifelong treatment. This study included Dr. Gatell of the Barcelona Clinical Hospital and Dr. Clotet of the Germans Trías i Pujol Hospital in Badalona.
The Clotet team investigates whether erraltegrabir can destroy HIV deposits to ensure that AIDS is possible thanks to such hopeful results. In fact, even if the level of the virus in the blood is not detectable, if HIV deposits are not destroyed, the virus could double again and it would be impossible to completely cure the patient. The Gatell team investigates how to reduce the side effects of the new drug.
In addition, it is important to note that antiretroviral therapies do not only aim to improve the health of the contaminated. Reducing the number of viruses, in addition to improving the health of the patient, reduces contagiousness and we usually do not take into account that another function of antiretroviral therapy is prophylaxis, since we associate prophylaxis only with the use of condoms or non-separation of syringes.
Dark vaccines
Among the antiretroviral measures, the most bitter is the vaccine. The isolation of HIV in 1983 allowed the development of a vaccine that would break the spread of the virus. Moreover, in a short time, when an effective vaccine against cat leukemia virus similar to HIV was achieved, researchers began to see closer the vaccine that would fight the pandemic. But they have not yet succeeded. In the last 25 years great efforts have been made and much money has been allocated to these investigations. In 2006, for example, $933 million was invested in HIV vaccine development projects.

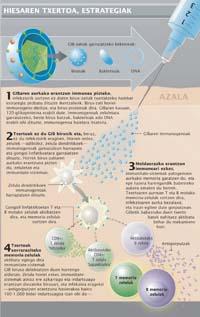
From the pharmacological point of view different strategies have been used for the development of the vaccine. Initially we searched for molecules that generate an adequate response of the immune system, that is, immunogens that increase the concentration of antibodies capable of neutralizing the virus. After intense research, the alleged ideal immunogen, the 120-glycoprotein surface of GIB-1, was discovered.
In this sense, the III Glycoprotein Vaccine 120-20. The results of two phase III clinical trials, the last pre-commercial phase, were published in 2005 and 2006. The results showed that the vaccine was able to increase the number of antibodies against 120-glycoprotein, but that it was not able to immunize healthy people against HIV. That is, people vaccinated with anti-glycoprotein 120-20 antibodies have the same chance of getting infected with HIV as non-vaccinated people. According to experts, the lack of preventive capacity of the vaccine is a consequence of the genetic variability of the virus and its ability to escape antibodies.
In view of the poor results of the immune-based vaccine, forces were deposited in type T lymphocytes. The aim of this second strategy, compared to the previous one, was not prophylactic. In this case, stimulating the response of type T lymphocytes was intended to reduce the initial number of infection viruses, which has shown slower disease development and less transmission of the virus.
In September 2007, the results of the clinical trial for 3,000 people --V520 from Merck and the U.S. Department of Health - were published with the first vaccine to increase the response of T lymphocytes. Unfortunately, according to the analysis of provisional results, although the vaccine increases the response of T cells, it is not able to reduce the number of initial viruses of the infection. However, the study results have shown the highest risk of HIV infection among vaccinated people.
For the moment, it seems that the future is dark. However, not all have yielded in the search for the protective vaccine. For example, a new phase 1 clinical trial of 16,000 men and women in Thailand is underway.
The first phase of this trial consisted of giving volunteers a T-type vaccine that activates more specific T-type lymphocytes than those that activated the V520, vCP1521 vaccine. In the second phase, they were given an antibody vaccine against glycoprotein 120-20. In this way, we have managed to group in the same trial the two therapeutic strategies investigated so far, on the one hand, activating T lymphocytes is a method of wrong attack and on the other hand, glycoprotein 120 of the surface of the virus is directly added. As research results are expected for 2009, we may be more optimistic within a year.


Gai honi buruzko eduki gehiago
Elhuyarrek garatutako teknologia




