Drug barrier race
2002/12/01 Galarraga Aiestaran, Ana - Elhuyar Zientzia Iturria: Elhuyar aldizkaria
Man has always sought substances that cure or relieve diseases. Initially they were mainly obtained from plants and animals, but since World War II, chemical synthesis and biotechnology have helped most new drugs. In
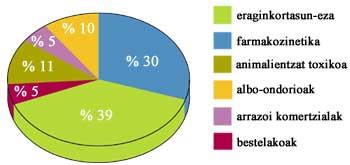
obtaining and marketing a new drug specialists intervene in areas as diverse as chemistry, molecular biology, basic and clinical pharmacy, economics and statistics. To the intellectual work we must add the money, since it is known that the pharmaceutical industries obtain enormous benefits thanks to a series of prestigious drugs, but it is also true that the investigations are expensive and last for many years. When a drug is marketed it can take between 10 and 15 years and research can cost between tens and hundreds of millions of euros.
Finding the motive molecule
The search for a new drug begins with the search for possible active components. The active component is the heart of the drug, with pharmacological effects. Before, most were obtained from plants: seeing that the effects of a plant on animals and humans were favorable, it was concluded that it served to cure it. Subsequently, an attempt was made to identify and isolate the active component of plants traditionally used in medicine, in order to synthesize it in the laboratory.
At the same time, new molecules with healing potential began to be performed and tested, but the search was blind, not based on the scientific method.
Currently, the search is based on knowledge of the mechanisms of the disease. Knowing that the disease causes biochemical and cellular changes, researchers try to look for substances that address these changes. It is about identifying the receptor and synthesizing the molecule that binds it. Thus, by associating this molecule to the receptor, it is possible to increase or inhibit the activity that the cell would have of itself.
On other occasions, the goal is to improve a molecule that is already known and to do this, structural changes are tested; sometimes, it is enough to slightly modify the structure to increase the efficiency considerably or make it suitable for another use.
It may also happen that we randomly find the active component of a new drug. This happened, for example, with penicillin. History is known: While Alexander Fleming worked with the flu virus, he discovered that in some bacterial cultures something impeded the proliferation of the bacterium Staphylococcus, a bacterial culture that was elusive and that the culprit was a mold substance. And so he discovered penicillin, the most widely used antibiotic for many years.
Claudio Palomo, professor at the Faculty of Chemistry of the UPV/EHU in Donostia, believes that instinct has a great importance in the search for new drugs. In fact, the Organic Chemistry section is testing a random molecule; although research is in the early stages, the results are very satisfactory.
However, not everything can be attributed to chance; Fleming's discovery took place in 1928, but in both 1875 and 1925 other researchers who wrote that mold had an antibacterial effect, but were not able to go deeper. So to find a new medicine, luck is not enough.
First tests

In all cases, the next step is to synthesize a few grams. They carry out the first tests of the candidate molecule. Molecules are often very effective in trials, but very few outperform tests prior to the start of clinical trials and, of course, fewer are at the end. According to US data, of the 5,000 compounds that begin to be studied in the laboratory, only five get tested in humans. And only one of these five is effective and safe enough to access the medical clinic.
The objective of the first tests is to know in detail the characteristics of the active component: physical-chemical characteristics, stability, solubility, etc. In addition, the influence of the component is analyzed in vitro through non-cellular systems (in enzymatic preparations) and animal or human cell cultures.
First steps at the Faculty of Chemistry of San Sebastian at UPV

In Euskal Herria, the university, hospital and laboratory work together to get new medicines. The Department of Organic Chemistry of the Faculty of Chemistry of the UPV/EHU is one of the participants in this work. This is where the first step is taken, that is, new active components are sought.
They have several search routes. A few years ago they developed their own peptide creation methodology. Peptides are molecules formed by 3-4 amino acids that, although small, can influence. One of these peptides is currently being studied. According to Professor Iñaki Gamboa, the process is not easy, but it is believed that if it goes well it is an important step.
Pharmacological drug
Another important step must be taken to make the drug with the drug. The drug and the drug are normally considered synonymous, but according to José Luis Pedraz, professor at the Faculty of Pharmacy of the UPV/EHU in Vitoria-Gasteiz, they are not the same: the drug is a molecule of pharmacological effect as the preparation to cure.

The work of pharmacists consists in the preparation of a preparation with which patients can be administered. The preparation, in addition to being ingested by one way or another, must meet other characteristics such as being stable, releasing the active component after administration and being able to act. All of this is not easy and you may need to do without an effective candidate molecule because you cannot prepare the medicine. Pedrat believes that one of the challenges of today's pharmacology is that genes or fragments of DNA and powerful molecules can become drugs. And, although they can be very useful to cure diseases, it is still not possible to take them as other medicines.
When the medicine is prepared with the motor molecule, toxicity tests in animals begin. The first sessions are done with the mouse, on the one hand because being small is not necessary large quantities and on the other, because there are regulated procedures in the mouse to measure toxicity. Later tests are done with larger animals.
These sessions analyze the interval between lethal and effective doses; the greater the interval, the safer the drug is. In addition, adverse effects are observed, both immediately and in the long term, as in future generations.
Measuring drug safety in humans
When the results of all the above tests are satisfactory, we must see what effect the drug has on humans. For this purpose, clinical sessions are conducted and the therapeutic effects and adverse effects are analyzed. Depending on the results, it is determined whether or not it serves therapy and, if valid, dose and dosage (when and how many) are determined.
Clinical sessions are divided into four phases according to the objectives. The main objectives of these four phases are, respectively, the analysis of safety, the measurement of efficiency, the comparison with other medicines and the monitoring and deepening of research once marketed.
In the first phase, therefore, the drug must be shown to be safe. The importance of this phase is enormous, as until then this substance has never been tested in humans. And if the results are not good, the investigation is immediately suspended. The tests are done with healthy volunteers and it should be noted that they are aware of what this medicine can cause and that they can abandon it when they want it.
At the beginning of clinical sessions the most difficult thing is to decide which dose to use. The starting point is animal experiments, with an initial dose of 1-2% of what is effective in animals and a maximum dose of 10-16% of the maximum allowed by animals. In the first studies a single dose is given, and after seeing what happens it is decided whether to give the following or not. Gradually the dose increases until unacceptable effects are detected.
With the least number of participants possible (30-100) it is intended to obtain the maximum possible information. To see how the body takes the medication, vital signs and heart activity are measured and blood and urine tests are done. Pharmacodynamic studies (drugs and their effects on the body) and pharmacokinetics (how it affects the body's metabolism to the drug) are then carried out and their interaction with other foods and/or drugs is observed. All this shows whether it is safe or not. Tests also help decide when and how to administer them.
Pharmaceutical Development
Unit of Vitoria-Gasteiz

The work started at the Faculty of Chemistry of San Sebastian could continue at the Faculty of Pharmacy of Vitoria-Gasteiz. Specifically, within the LEIA Foundation, the Pharmaceutical Development Unit is formed by the Department of Pharmacy and Pharmaceutical Technology and the Clinical Trials Unit of the Txagorritxu Hospital.
In the Faculty of Pharmacy they study candidate molecules and define their characteristics. If you see that a molecule is valid, prepare the medicine. Subsequently, the Clinical Trials Unit of Txagorritxu Hospital holds sessions with volunteers from the first phase. Blood and urine tests that should be done are also done in this section of Pharmacy. If everything goes well they go to the second phase of the clinical sessions, but for this they need the collaboration of other hospitals.
In addition, generic medicines are prepared and tested in Vitoria-Gasteiz, some of which are marketed through the Litaphar de Azpeitia laboratory.
Sure yes, but... is it effective?

A second phase analyzes the healing of the drug. Until then only safety has been looked at, but no condition does not mean you benefit. In a second phase tests are carried out to check the possible beneficial effect and determine the most appropriate rules and doses for the administration of the drug.
The second phase lasts between one and three years and requires the participation of between 100 and 400 people, some of them patients. Finally the dose-effect ratio is known. In the sessions there is always a control group and the tests are usually random and blind, that is, the participant does not know if he is taking the medicine or placebo. Sometimes blind double sessions are held, that is, the researcher who gives medicine to participants does not know what he is taking either. In this way, the results are more objective.
Last pre-commercial phase
The third phase gathers the latest tests done before marketing the drug. In this phase the drug is compared with other drugs already existing in the market and of equal therapeutic use. In addition, the tests are done as if the patients were in normal situation, that is, they should take the dose at the same time and in the same state in which they should take the medication, continue with normal activity and food...

Do not forget that the medicine is aimed at patients. Therefore, third phase volunteers should replace patients who will receive the medication as best as possible. However, it is not always easy, since testing with sick, vulnerable, children, elderly and pregnant causes ethical problems.
These teams are more vulnerable than the rest, so some researchers exclude them from testing. Overprotection, however, is usually not beneficial because the population excluded from trials, if one day you need this medicine, no one will know what its effect will be.
Apart from this, it is necessary to be very clear what you want to get in the third phase, how much time and how much money you want to spend. In fact, many new drugs are more effective than existing ones, but we have to see to what extent the difference is so great that it is often not very large. In these cases, it may be better to suspend the investigation, since the difference does not deserve so much money and time.
After the dismissal we must continue investigating

With all the information collected in the first three phases of the clinical sessions, marketing authorization application reports are prepared. Considering where you want to market, reports are sent to the Spanish Medicines Agency, the European Agency or the U.S. Food and Drug Administration (FDA). so that, after a thorough analysis, they authorize or deny dismissal.
But when deciding, a lot of time is often spent, as they often ask for data completion or further testing. According to Lidia Larrañaga, technical director of the laboratory Litaphar de Azpeitia, in the case of generic drugs this process takes about a year. Logically, when the drug is completely new, the process of obtaining the authorization lengthens a lot.
Once authorized, the drug goes on the market. This is when the fourth phase of clinical sessions begins. The objective of this phase is to monitor the efficacy and safety of the drug. It often happens, for example, that the established doses are too high, so it is republished with lower doses. To a lesser extent, serious side effects may appear in some group of the population that were not detected in clinical sessions. This can force the medicine to be removed from the market.
Other times, thanks to the studies carried out in this phase, new uses are found. For example, aspirin has been on the market for 105 years. At first it came out to eliminate pain and treat inflammations, but now it is known that it is also beneficial to prevent heart attacks and tests are being conducted to use it in the fight against cancer.
It is clear that the launch of a new drug is a painful, long and expensive work. In return, there are remedies against many diseases. However, medication is sometimes abused and the measurement depends on the doctor and everyone.
The future begins today
XX. The pharmacology of the twentieth century advanced greatly thanks to chemistry, pharmacology and clinic. From now on, experts believe that advances will focus on molecular biology, genome and proteome, biotechnology, combinatorial chemistry, bioinformatics and robotics.

Undoubtedly, the identification of genes has meant a new way of understanding diseases. Knowing the genetic control of cell functions, new ways to prevent or cure diseases are opened. Regardless of gene therapy, knowledge of the genome leads to discovering the fate of new drugs. Proteomics, for its part, analyzes the product of the genome, so it complements the previous one.
On the other hand, there are differences between ethnic groups when it comes to metabolizing medicines, and it is possible that a medicine that benefits most may cause harm to some. Pharmacogenetics studies the relationship between individual genotype and drug metabolization capacity.
Combinatorial chemistry has been a breakthrough in synthesis chemistry. The current technology allows to obtain molecules of interest faster than ever. To know which of these molecules are actually of therapeutic value, rapid screening methods of high efficacy are used. Thus, the distinctives between 50,000 and 60,000 molecules per week are known with precision and rigor. All this is due to bioinformatics as an essential tool for the quick understanding and treatment of so many data.
However, natural substances with curative capacity should not be avoided. So far only 10% of Earth's resources have been exploited, so many hidden treasures remain. To be aware of its importance, it is enough to see which are the best-selling drugs, such as cyclosporine, lobastatine, clarithromycin, pravatatin and altobastatine, nine of the ten non-protein drugs that were sold last year in the US.

Gai honi buruzko eduki gehiago
Elhuyarrek garatutako teknologia